While the commercial market is moving toward rechargeable power sources, military users are consuming the primary power sources (non rechargeable batteries) due to the simple logistics, long shelf life, readiness and robustness. Performance requirements for military grade portable power sources are much more demanding than commercial batteries. Military equipment requires high power rate and light weight (more power per volume unit). Weight consideration is a critical aspect, especially for dismounted operations. Military specs are also more demanding in the operating temperature and humidity range, water and salt resistance, as well as safety. These requirements limited the variations of primary battery chemistries and designs. In recent years, several armies have shifted to the use of rechargeable batteries for peacetime, training and garrison operations, as well as specific combat uses, to save in cost and transportation.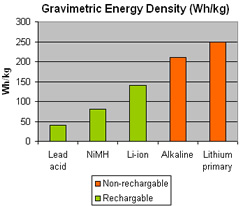
The consideration for primary batteries is clear – combat readiness require immediate response, therefore, immediate availability of full and consistent power, no charging and priming before use – dictates the use of primary batteries that have no voltage delays, even after long storage periods. The implementation of modern lithium batteries significantly increased power density and reduced weight, when compared to earlier alkaline and carbon-zinc technology. One such technology is the matured Lithium sulfur dioxide (Li/SO2) primary battery technology. However, one of the major concerns for military users was the pressurized cylinders that compose the Li/SO2 cell. These cylinders contain electrolytes stored at high pressure that can explode if punctured by enemy fire or physical abuse. New production processes of Li/SO2 batteries have utilized non-pressurized cells. For example, a lithium-manganese dioxide (Li/MnO2) cell is constructed as a laminated, aluminized pouch that offers high energy density. Another concern was the emission of hazardous gases, in case of short circuit or overheating. Modern designs, especially those used in night vision equipment, are now being replaced with Li/MnO2 cells that have safety features to automatically shut down of the battery, when overheated. Modern portable electronic equipment is now moving toward the new Li/MnO2 chemistry, which has higher energy density than Li/SO2, since the cell utilizes the entire battery cavity.
Additional Parts of this article :
- Powering the Land Warrior
- Military use of Primary Lithium Batteries
- Rechargeable Power For Military Use
- Logistic Aspects of Military Batteries
- Logistics of Military Rechargeable Battery



















